Talk to us
Diphtheria Tetanus And Pertussis Vaccine
MOQ : 50 Packs
Diphtheria Tetanus And Pertussis Vaccine Specification
- Salt Composition
- Diphtheria, tetanus, pertussis (acellular)
- Packaging Type
- Box
- Life Span
- As per expiry date
- Dosage Form
- Injection
- Origin of Medicine
- Gsk
- Pacakaging (Quantity Per Box)
- 3
- Brand Name
- Infanrix
- Indication
- Prevention of diphtheria, tetanus and pertussis
- Drug Type
- Vaccine
- Ingredients
- Diphtheria toxoid, Tetanus toxoid, Acellular pertussis components
- Physical Form
- Liquid Suspension
- Function
- Immunization
- Recommended For
- Infants and children
- Dosage
- As per physicians advice
- Dosage Guidelines
- Intramuscular injection
- Suitable For
- Children
- Quantity
- 3
- Storage Instructions
- Store at 2C-8C, do not freeze
- Shake Well
- Before use
- Volume Per Dose
- 0.5 ml
- Route of Administration
- Intramuscular
- Color
- Clear
- EU Pharmacopoeia
- Compliant
- Prescription Required
- Yes
- Multi-dose Vial
- No
- Stabilizer
- Contains aluminium salt
- Age Group
- Paediatric use
- Expiry Status
- Clearly labelled
Diphtheria Tetanus And Pertussis Vaccine Trade Information
- Minimum Order Quantity
- 50 Packs
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 2-10 Days
- Main Domestic Market
- All India
About Diphtheria Tetanus And Pertussis Vaccine
Introducing Infanrix, a top-tier diphtheria, tetanus, and pertussis vaccine for paediatric use, developed by the renowned GSK. This clear liquid suspension ensures excellent immunization efficacy. Each 0.5 ml intramuscular dose contains rare acellular pertussis components, stabilized with aluminium salt, providing reduced reactogenicity. With EU Pharmacopoeia compliance and expiry status clearly labelled, Infanrix offers sizzling peace of mind for parents and clinicians alike. Supplied as a single-dose, non-multidose vial, this superior vaccine is indicated for the prevention of serious infections in infants and children. Storage is straightforward at 2-8C. Prescription required.
Commercially Relevant Vaccine for Paediatric Protection
The Diphtheria Tetanus and Pertussis Vaccine is highly valued in pediatric healthcare, offering superb protection against three life-threatening diseases. Regularly used in immunization programs, clinics, and hospitals, its usage type is strictly prescribed for infants and children, ensuring top-ranking safety and efficacy. The vaccine is injected intramuscularly according to physician's advice, providing consistent results for commercial health suppliers, pediatricians, and public health authorities.
Certifications, Sample Availability, and Reliable Supply Outlay
This vaccine boasts EU Pharmacopoeia valuation and a well-documented certification record, assuring healthcare professionals of its premium quality. Samples may be available for evaluation to streamline commercial outlay decisions. Reliable delivery timelines are maintained, with supply originating in India via authorized traders and suppliers. Each transaction is supported by clear packaging, labelled expiry status, and robust cold chain logistics to guarantee top-tier preservation from dispatch to point-of-use.
Commercially Relevant Vaccine for Paediatric Protection
The Diphtheria Tetanus and Pertussis Vaccine is highly valued in pediatric healthcare, offering superb protection against three life-threatening diseases. Regularly used in immunization programs, clinics, and hospitals, its usage type is strictly prescribed for infants and children, ensuring top-ranking safety and efficacy. The vaccine is injected intramuscularly according to physician's advice, providing consistent results for commercial health suppliers, pediatricians, and public health authorities.
Certifications, Sample Availability, and Reliable Supply Outlay
This vaccine boasts EU Pharmacopoeia valuation and a well-documented certification record, assuring healthcare professionals of its premium quality. Samples may be available for evaluation to streamline commercial outlay decisions. Reliable delivery timelines are maintained, with supply originating in India via authorized traders and suppliers. Each transaction is supported by clear packaging, labelled expiry status, and robust cold chain logistics to guarantee top-tier preservation from dispatch to point-of-use.
FAQ's of Diphtheria Tetanus And Pertussis Vaccine:
Q: How should the Diphtheria Tetanus And Pertussis Vaccine be administered?
A: The vaccine should be administered as an intramuscular injection by a healthcare professional, following the physician's recommended schedule for infants and children.Q: What are the key benefits of using Infanrix for immunization?
A: Infanrix offers excellent protection against diphtheria, tetanus, and pertussis, with reduced side effects thanks to its acellular composition and rare, high-grade formulation, compliant with EU standards.Q: When is this vaccine indicated for use?
A: This vaccine is recommended for immunization of infants and children to prevent diphtheria, tetanus, and pertussis as per pediatric vaccination schedules.Q: Where should the vaccine be stored to maintain its efficacy?
A: The vaccine must be stored at temperatures between 2C and 8C; freezing should be avoided to maintain potency and effectiveness.Q: What is the process for ensuring safe usage and handling?
A: Shake the vial well before use, verify the expiry date, and confirm dosage with the healthcare provider. Proper refrigeration and single-use injection practices are essential for ensuring safety and quality.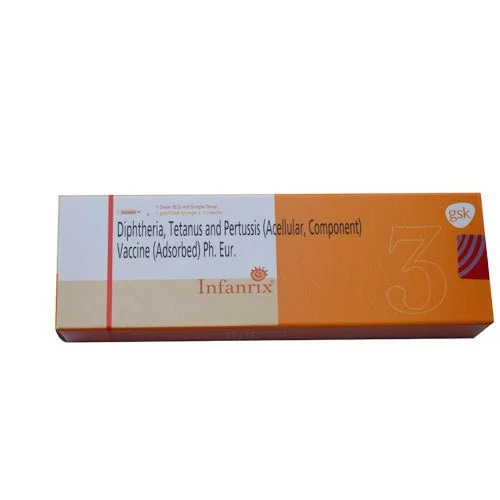
More Products in Pharmaceutical Vaccine Category
Rotavirus Vaccine IP
Minimum Order Quantity : 50 Packs
Origin of Medicine : India
Packaging Type : Vial with dropper
Dosage : As directed by physician, typically 1.5 ml per dose
Life Span : Up to 36 months from date of manufacture
Dosage Form : Oral suspension
Pneumococcal Polysaccharide Conjugate Vaccine
Minimum Order Quantity : 50 Packs
Origin of Medicine : India
Packaging Type : Vial with syringe
Dosage : 0.5 mL per injection
Life Span : 24 months from manufacturing date
Dosage Form : Injection
Heptatis BB Vaccine IP
Minimum Order Quantity : 50 Packs
Origin of Medicine : Indian
Packaging Type : Vial with sterile syringe
Dosage : As prescribed by the physician, usually 0.5 ml for children and 1 ml for adults
Life Span : 36 months from the date of manufacture
Dosage Form : Injection
0.5 ML Hepatitis-B Vaccine IP
Minimum Order Quantity : 50 Packs
Origin of Medicine : India
Packaging Type : Glass vial with rubber stopper
Dosage : 0.5 ml per dose
Life Span : 36 months from date of manufacture
Dosage Form : Injection
 Send Inquiry
Send Inquiry